By Soyeon Park
Note: this article does not constitute medical advice, and serves merely as an exploration of a scientific concept. This article does not seek to promote any specific medication.
Eggs are a simple ingredient, but they can be cooked into various foods such as omelets, fried eggs, or boiled eggs, with very different tastes and textures based on how they are processed. Similarly, in chemistry, two molecules with the same atoms can be completely different compounds that have different properties due to only slight differences in their structures. These molecules are called isomers.
In pharmacy, isomers are particularly important because small differences in the properties among isomers can mean the difference between life and death [1]. Isomers can be separated into two groups, including structural isomers, in which the atoms are bonded differently, and stereoisomers, in which the atoms are bonded to the same atoms but arranged differently in three dimensions [7]. Among these stereoisomers, the enantiomers, which are also called mirror isomers, are extremely remarkable.
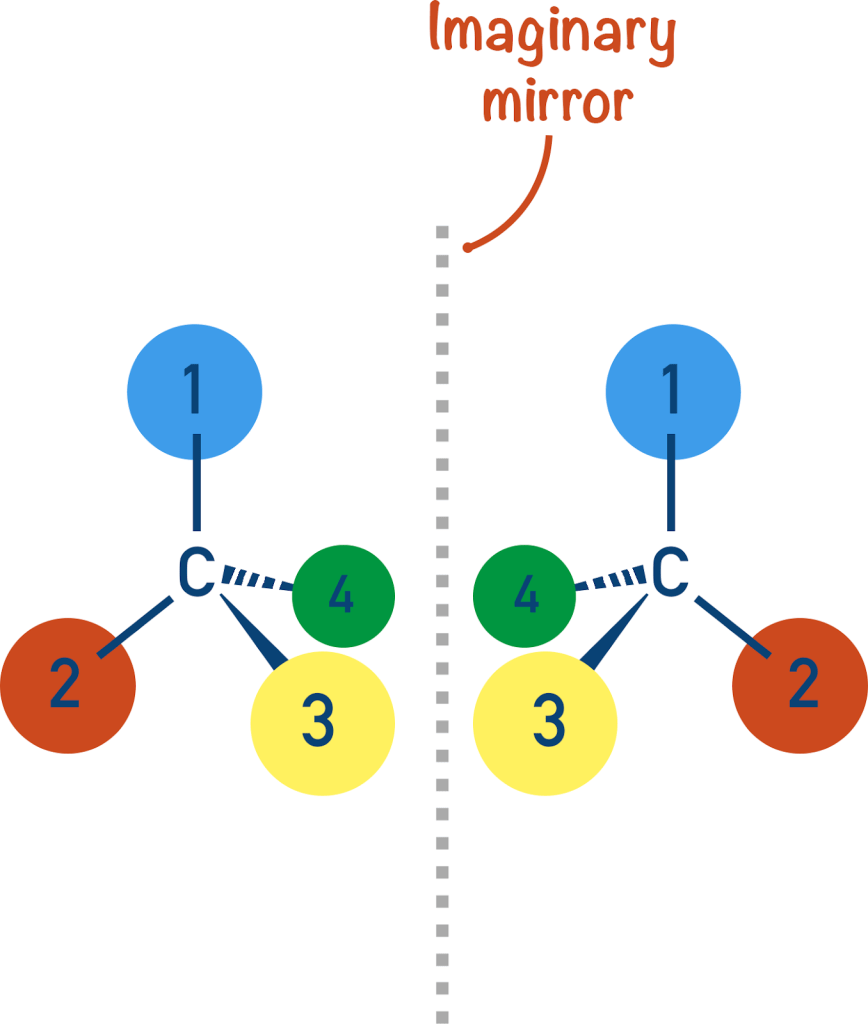
Figure 1. Image of the enantiomers [7]
As can be seen from the diagram, the compounds on the right and left appear very similar to one another but are actually mirror images; this means that they cannot be superimposed (essentially placed on top of each other and be identical). While this seems to be nothing more than a shape difference, it has great implications in biological systems [7]. Because enzymes and receptors in our bodies also have specific orientations, a different orientation of a molecule can result in a completely different type of functioning, or no functioning at all. That is, one molecule can be the “key,” while its stereoisomer is not [2].
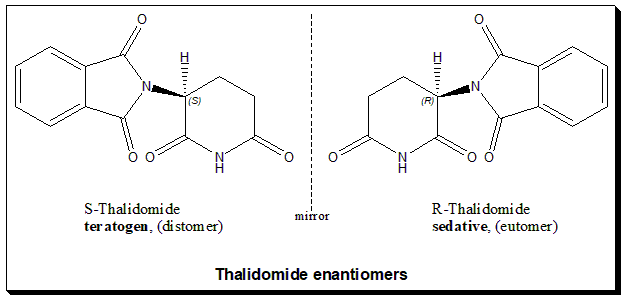
Figure 2. Image of Thalidomide enantiomers [8]
Figure 3. Image of thalidomide [9]
A notable case in point of what small differences in molecules can do is thalidomide. Thalidomide was promoted in the late 1950s as a medicine for alleviating morning sickness during pregnancy [3]. However, an ignorance of enantiomers shortly turned thalidomide into one of medicine’s worst tragedies. While one of thalidomide’s enantiomers was an anti-nauseant, the other caused lethal birth defects. Having no idea that they were different, the two forms were sold as a single drug, and many mothers took the damaging enantiomer. This resulted in thousands of babies being born with short limbs, missing fingers, and other defects, including those in Europe and the rest of the world [3].
The thalidomide horror exposed the profound lack of consideration for molecular structure in medicine at the time, shaking the medical establishment to its core and bringing about radical changes in drug testing, approval, and regulation [4]. This episode thus began the modern clinical trial system, requiring rigorous safety testing of all chemically active drugs before they are released into the public domain. Following the thalidomide disaster, pharmaceutical researchers began to rigorously control molecular handedness, or chirality, from the drug design stage [2].
The result of this rigorous control is esomeprazole, a drug prepared by selecting only the left-handed enantiomer of omeprazole, which is used to treat gastroesophageal reflux disease (GERD). The original omeprazole was an R-S racemic mixture. However, the two types were different in terms of absorption rates and efficiency in the body, and they therefore resulted in unstable efficacy in some patients or side effects in liver metabolism. In order to enhance efficacy and reduce side effects, esomeprazole was created by refining only the S-form [6]. This version was absorbed stably with minimal side effects and produced a long-lasting gastric acid suppression effect [5]. That is, enantiomer separation produced improved results and decreased side effects.
Bibliography:
- Chhabra, N., Aseri, M. and Padmanabhan, D. (2013). A review of drug isomerism and its significance. International Journal of Applied and Basic Medical Research, [online] 3(1), p.16. doi:https://doi.org/10.4103/2229-516x.112233.
- Senkuttuvan, N., Komarasamy, B., Krishnamoorthy, R., Sarkar, S., Dhanasekaran, S. and Anaikutti, P. (2024). The significance of chirality in contemporary drug discovery-a mini review. RSC Advances, [online] 14(45), pp.33429–33448. doi:https://doi.org/10.1039/d4ra05694a.
- Grogan, D.P. and Winston, N.R. (2021). Thalidomide. [online] PubMed. Available at: https://www.ncbi.nlm.nih.gov/books/NBK557706/
- Vargesson, N. (2015). Thalidomide-induced teratogenesis: History and mechanisms. Birth Defects Research Part C: Embryo Today: Reviews, [online] 105(2), pp.140–156. doi:https://doi.org/10.1002/bdrc.21096.
- Pai and Pai N (2007). Recent advances in chirally pure proton pump inhibitors. PubMed, 105(8), pp.469–70, 472, 474.
- Tonini, M., Vigneri, S., Savarino, V. and Scarpignato, C. (2001). Clinical pharmacology and safety profile of esomeprazole, the first enantiomerically pure proton pump inhibitor. Digestive and Liver Disease, 33(7), pp.600–606. doi:https://doi.org/10.1016/s1590-8658(01)80115-8.
- http://www.chemistrystudent.com. (n.d.). Optical Isomerism (A-Level) | ChemistryStudent. [online] Available at: https://www.chemistrystudent.com/opticalisomerism.html.
- Anon, (n.d.). Thalidomide – Chiralpedia. [online] Available at: https://chiralpedia.com/blog/thalidomide/.
- Science Museum (2019). Thalidomide. [online] Science Museum. Available at: https://www.sciencemuseum.org.uk/objects-and-stories/medicine/thalidomide.


Leave a comment